Cookies & Your Privacy
This website uses cookies to ensure you get the best experience on our website. More Information >

Accu-Steel uses a variety of heavy-duty and high-quality materials to create the best fabric covered storage buildings, including inline and hot-dipped galvanizing techniques.
By: Philip G. Rahrig, Executive Director, American Galvanizers Association
The two most common methods of applying zinc metal to steel handrail tubing for the purpose of protection from corrosion are batch hot-dip galvanizing and in-line, continuous galvanizing.
Batch hot-dipped galvanizing involves loading a rack or overhead lifting fixture with 50 or more cut-lengths (usually 20’ or 40’) of tubing, thoroughly cleaning the steel, and then immersing the entire load into a molten zinc bath where all exterior and interior surfaces are thoroughly coated. Once the newly galvanized tubing cools, it is ready for shipment to the job site or transport to a paint contractor for application of paint.
In-line, continuous galvanizing involves feeding the tubing through the bath of molten zinc and then applying a conversion coating to prevent the formation of naturally occurring zinc oxide and hydroxide. Usually, a topcoat of a clear, inorganic, polymeric paint is applied to the conversion coating. It is important to note that only the outside of the tubing goes through this process; the tubing’s inside surface receives only a coating of zinc-rich paint.
Fundamentally, zinc metal will provide some level of corrosion protection to the steel used in myriad applications. However, understanding the metallurgy, bond strength, corrosion mechanisms, and testing of the two methods described above may lead to better design decisions for particular applications.
Zinc Coating Thickness and Service Life
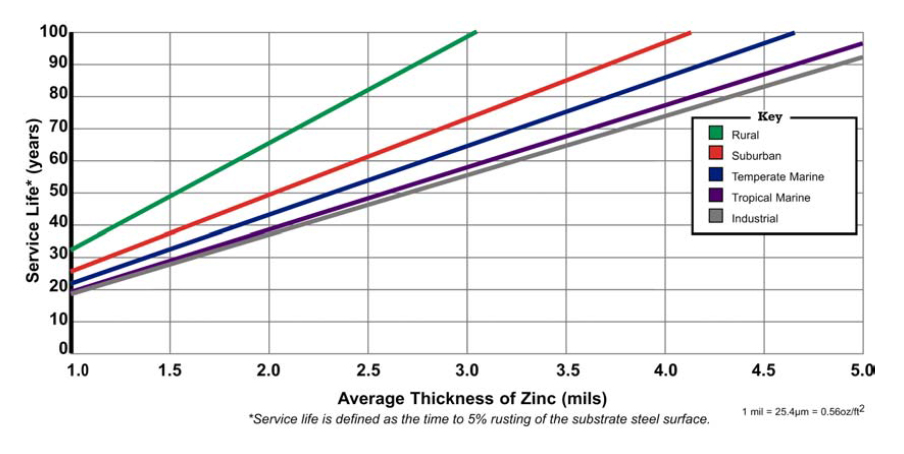
On standard 0.25” wall tubing, the batch hot-dip galvanizing process applies a minimum of 3.0 mils (1.7 oz/ft2) of protective zinc. The in-line process applies about 0.9 mils (0.5 oz/ft2). As the “Hot-Dip Galvanized Service Life” chart (Fig. 1) indicates, the batch-process-produced zinc coating will perform in use, without maintenance, for approximately three times longer than the in-line-produced zinc coating.
Taking into consideration the organic clear-coat applied to the handrail galvanized in the continuous process, it is important to note that applying a barrier protection system over a galvanized coating (by whichever process) results in service 1.5 times the sum of the service life of the two systems. While applying a barrier coating to the in-line galvanized handrail will enhance the service-life, it still does not match that of the unpainted batch-process galvanized handrail. And, as the interior of the continuous-process handrail is not provided a metallurgically bonded zinc coating, it is difficult, at best, to accurately compare service-life expectancies.
Bond Strength
Batch hot-dip galvanizing process generates a metallurgical reaction between the molten zinc and the iron in the steel tubing. This reaction results in the formation of three zinc-iron alloy layers metallurgically bonded to the base metal, topped by an impact-resistant pure zinc outer layer. The metallurgical bond between the zinc-iron alloy layers and the steel is measured at approximately 3600 psi. This bond strength is especially important for the interior surface of the batch-galvanized tubing, where the coating resists corrosion caused by trapped water and moisture. In comparison, the zinc-rich paint applied to the inside of tubing galvanized via the in-line, continuous process has a mechanical bond in the range of 300 – 500 psi. Because of this lesser bond strength, it is possible for trapped moisture to make its way between the zinc-rich paint and the steel tubing, causing rust formation and eventual flaking and failure of the paint system.
Corrosion Protection
Batch hot-dip galvanizing metallurgically bonds zinc to all surfaces of the tubing. In fact, batch galvanized tubing and pipe are often used in fabrications with hot-dip galvanized vessels and tanks to store a wide range of liquids. The zinc-rich paint applied to the inside of tubing galvanized via continuous in-line processes provides some corrosion protection, but performs less effectively when exposed to liquids. In such cases, moisture makes its way to the base steel of the tubing through cracks, damaged areas or porosity in the zinc-rich paint, allowing corrosion to begin where it is not apparent and is not easily remedied.
Inspection and Testing
Salt-spray or salt-fog tests are often used to compare the corrosion protection provided by various coatings. Simply put, galvanized coatings in use require wet and dry cycles in order to develop a naturally occurring, protective zinc-carbonate patina that increases the coating’s long-lasting corrosion protection. It is widely understood that laboratory salt tests are not reflective of real-world situations. So, it is unrealistic and ill-advised to use salt spray tests to develop a realistic comparison between batch hot-dip galvanized tubing (without an additional barrier protection system) to in-line galvanized tubing (that is additionally conversion coated and top-coated with inorganic polymeric paint).
Durability
The three-plus MLs of zinc in the zinc-iron alloy layers applied via the batch hot-dip galvanizing process is actually harder (250 DPN) than the base steel (159 DPN). It is extremely difficult to damage the thick zinc coating to the extent that corrosion protection would be affected. In comparison, the inorganic polymeric paint covering the thin layer of zinc applied in the in-line process damages as easily as any other paint and is particularly susceptible to deterioration caused by the sun’s ultraviolet rays. Any damage to the paint covering exposes the thin layer of zinc, resulting in a protection system that can be anticipated to last only as long as the zinc thickness will allow, or about a third as long as the batch galvanized tubing. Additionally, surface contaminants and normal in-use wear and tear are particularly threatening to topcoat paints.
Touchup and Repair
Batch hot-dip galvanizing is done after the tubing is cut to length and protects all surface areas. Touchup is rarely required after transport and field construction because the thick zinc coating is hard and bound tightly to the steel surface. In-line continuous galvanized tubing is cut to lengths after galvanizing. These unprotected ends are susceptible to corrosion if not touched up with zinc solder, metalizing spray, or zinc-rich paint, and will be potential sites of structural failure.
Making a Choice
Clearly, an architect’s or engineer’s decision to use batch hot-dip galvanized tubing or in-line continuously galvanized tubing depends on the design life desired for a handrail project and the funds available for future maintenance. While hot-dip galvanizing has advantages in service life, bond strength, corrosion protection, and durability; inline galvanizing may offer an initially more affordable coating alternative for applications in milder environments and projects that have lower bond strength requirements.
©2002 American Galvanizers Association. The information provided herein has been developed to provide accurate and authoritative information about after-fabrication hot-dip galvanized steel. This material provides general information only and is not intended as a substitute for competent, professional examination and verification as to suitability and applicability. The provided herein is not intended as a representation or warranty on the part of the American Galvanizers Association. Anyone making use of this information assumes all liability arising from such use.
Let the experts at Accu-Steel dispel any myths that you have been told about hot-dip galvanizing. With years of education and experience, our company has the knowledge you need to know about this method. Be sure to contact us today with any questions that you have. We are happy to help!
This website uses cookies to ensure you get the best experience on our website. More Information >